For centuries, the substances going with the dynamic pharmaceutical fixing (API)—known as excipients—were considered insignificant fillers, folios, or stabilizers. Their essential part was to give bulk, keep up keenness, and guarantee understanding compliance. Nowadays, in any case, these “dormant fixings” are anything but detached. They are presently seen as utilitarian, crucial enablers of next-generation treatments, from complex biologics to exceedingly focused on conveyance frameworks. The advancing part of novel excipients is not fair a logical reference; it’s a basic unused chapter in advanced medication, driving helpful development and challenging administrative ideal models worldwide.
From Antiquated Added substances to Useful Polymers: A Chronicled Context
The utilize of excipients dates back to old times, with early medicines found in Sumerian and Egyptian writings utilizing normal substances like nectar, starches, and plant extricates to make medications agreeable and administrable. This discernment of excipients as straightforward carriers held on for centuries.

A significant move started in the mid-20th century. As pharmaceutical innovation progressed, the industry realized that excipients were not inactive. High-profile occurrences, such as the alter of a diluent in a common epilepsy sedate that driven to a spike in poisonous quality, illustrated their significant affect on a drug’s bioavailability and discharge profile. This acknowledgment impelled a more profound logical examination and the foundation of comprehensive administrative guidelines. The improvement of manufactured and semi-synthetic excipients, like progressed celluloses and polymers, stamped the genuine starting of their useful time, permitting formulators exceptional control over a dose form’s performance.
The Unused Wilderness: Why Oddity is Necessary
The require for novel excipients has quickened due to the challenges displayed by cutting edge sedate candidates. Today’s APIs including little atoms with destitute dissolvability or porousness, and huge biologic particles like proteins, peptides, and nucleic acids often show noteworthy definition obstacles that routine excipients essentially cannot solve.

Current Patterns and Innovation
Novel excipients are presently designed to perform specialized, high-impact capacities, changing the exceptionally design of medicate conveyance systems:
- Solubility and Bioavailability Upgrade: A tremendous number of unused sedate candidates are ineffectively solvent in water, extremely constraining their assimilation by the body. Novel excipients, such as self-emulsifying sedate conveyance frameworks (SEDDS), co-processed materials, and specialized polymers for nebulous strong scatterings, are fundamental for dissolving and stabilizing the API to definitely increment the sum of sedate that comes to the bloodstream.
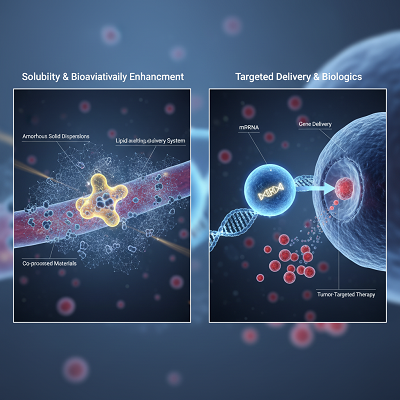
- Targeted Conveyance and Biologics: For complex particles like the mRNA in COVID-19 antibodies, novel excipients are the linchpin of victory. Lipid Nanoparticles (LNPs), a lesson of novel excipients, serve as defensive, utilitarian vehicles that shield the fragile mRNA from debasement and empower its exact conveyance into target cells. Essentially, biodegradable polymers are being utilized to make nanoparticles for cancer treatments, guaranteeing the medicate is discharged as it were at the tumor site.
- Controlled and Supported Discharge: Progressed polymers and specialized networks permit for medicate discharge to be absolutely tweaked over time. This usefulness is basic for long-acting injectables and inserts, which can essentially make strides understanding compliance and restorative results by decreasing dosing frequency.
- Abuse Discouragement: In the battle against the opioid emergency, novel excipients are key components in abuse-deterrent definitions, planned to make the medicate troublesome to pulverize, break down, or inject.
The Administrative Barricade: A Cautionary Stance
Despite their basic significance, the selection of novel excipients faces a noteworthy administrative jump. As a few master overviews have affirmed, the essential challenge is the need of an free, harmonized administrative pathway for excipients.

- The Master View:
“The industry is hesitant to take on the chance. Beneath the current framework, a novel excipient is regularly looked into as it were inside the setting of the last medicate application,” clarifies one detailing researcher. “In the event that the controller raises security concerns approximately the excipient late in the survey handle, the whole drug—on which millions have been spent—can be rejected or seriously delayed.”
Regulators like the U.S. Nourishment and Medicate Organization (FDA) frequently consider an excipient “novel” if it’s utilized for the to begin with time, at a higher dosage, or by means of a modern course of organization. This implies producers must supply comprehensive toxicological and quality information, comparative to that required for an API. This included time, fetched, and chance acts as a effective disincentive for both excipient producers and sedate engineers to improve past the existing, affirmed list of excipients.
In reaction, administrative bodies, counting the FDA, have propelled pilot programs to audit the quality and security of certain novel excipients exterior of the last medicate application. Partners broadly concur that a clear, committed administrative pathway is fundamental to open advancement in this field.
Implications for the Future of Medicine
The part of novel excipients will as it were develop as the complexity of medicate particles proceeds to rise. Their fruitful integration has significant suggestions for the pharmaceutical landscape:
- Enabling Neglected Needs: Novel excipients are the key to defining drugs that were already “undevelopable” due to destitute physicochemical properties. Without them, endless possibly life-saving therapeutics would never reach the patient.
- Personalized Medication: Future excipients may be outlined to react to patient-specific natural signals (e.g., pH, temperature, or protein action), clearing the way for genuinely shrewdly, personalized medicate delivery.
- Manufacturing Modernization: Excipients custom fitted for progressed fabricating strategies, such as ceaseless fabricating and 3D printing, will streamline generation, upgrade quality control, and eventually decrease costs.
The days of seeing excipients as inactive components are long over. They are presently recognized as modern drug-delivery foundation. For the pharmaceutical industry to keep pace with logical revelations in zones like quality treatment and nanotechnology, overcoming the administrative dormancy and cultivating strong development in the excipient space is no longer optional—it is basic for the future of persistent care.